Last Friday, global health and personal care products giant Johnson & Johnson announced a voluntary recall of about 33,000 baby powder bottles in the United States from a single 2018 lot after sub-trace levels of asbestos were found in one sample bottle purchased online and tested by the FDA.
Asbestos is a known carcinogen with no safe levels of exposure.
Johnson & Johnson advised consumers to discontinue use of baby powder from lot #22318RB. The lot number is located on the back “neck” of the bottle. Consumers can contact the company’s Consumer Care Center at www.johnsonsbaby.com or call 1-866-565-2229 for baby powder refund information.
Johnson & Johnson was notified on Friday by the FDA about the asbestos in its baby powder and issued the recall “out of an abundance of caution,” according to a company news release. The company stated that it had “immediately launched a rigorous, thorough investigation” into the matter, which could take 30 days or more.
In February, Johnson & Johnson disclosed that the Justice Department and the Securities and Exchange Commission were investigating allegations about asbestos-contaminated baby powder. However, this is the first time Johnson & Johnson has recalled baby powder due to asbestos concerns and the first time government regulators have found asbestos in the powder, Reuters reported.
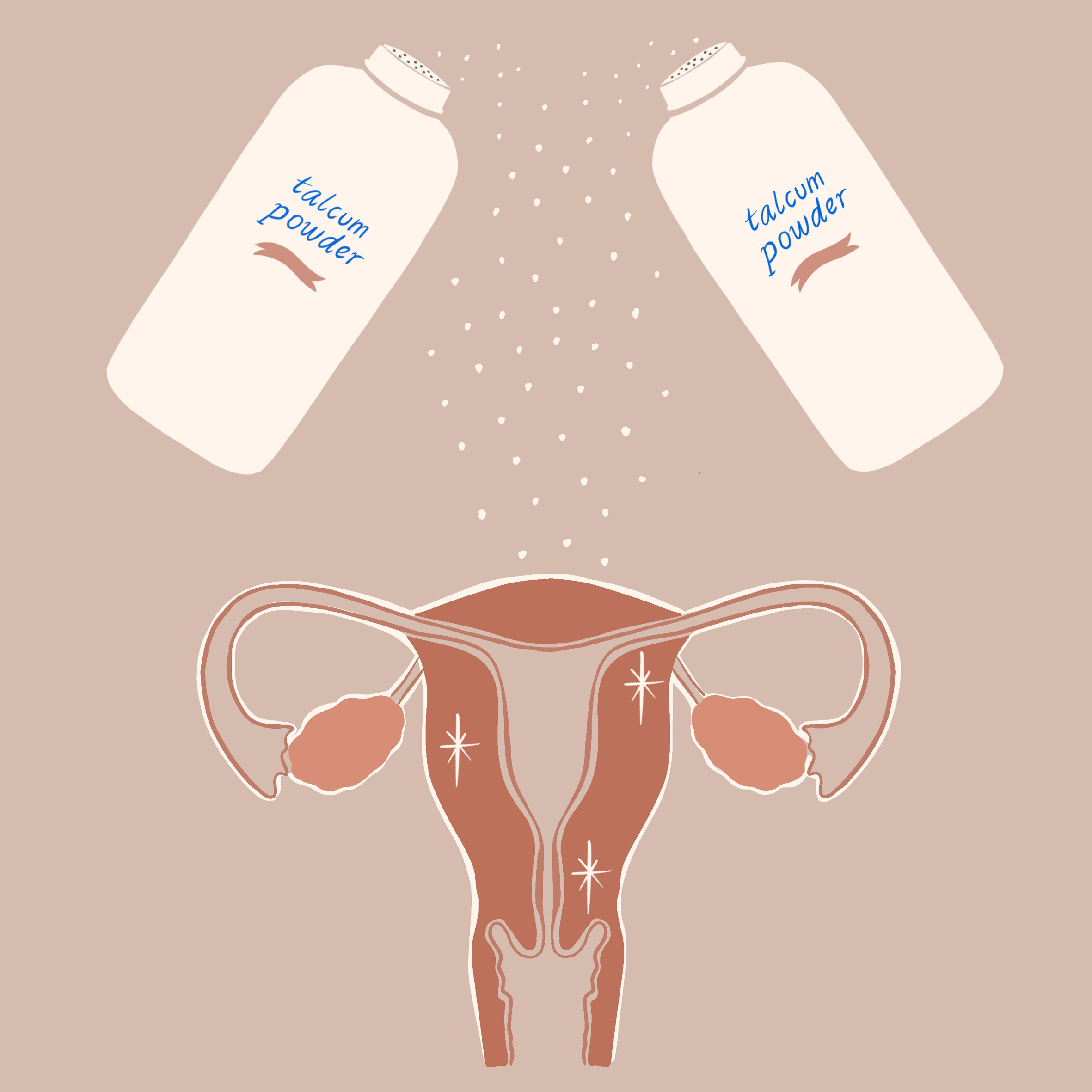
The timing for this incident could not be worse for Johnson & Johnson, which currently faces more than 15,000 lawsuits from consumers alleging that asbestos in the company’s baby powder caused their ovarian cancer or mesothelioma.
Mesothelioma is a deadly cancer of the mesothelium, the thin layer of tissues surrounding most internal organs. Ovarian cancer is likewise considered quite lethal – there’s no screening test and it often goes undetected until it’s too advanced to treat effectively.
Baby, or talcum powder, is made from the mineral talc which is mined and often naturally occurs in close proximity to asbestos, another mineral. When asbestos fibers are released into the air, they can enter a person’s body and become trapped in the lungs or in the digestive tract if swallowed. The body cannot break down these fibers once they are stuck.
In July 2018 a St. Louis jury ordered Johnson & Johnson to pay $4.69 billion in damages to 22 women who alleged that their ovarian cancer was caused by exposure to the company’s baby powder. The company is appealing this and other judgments.
Casting doubt on the validity of the government’s findings, Johnson & Johnson stated that it is as yet unable to confirm or deny whether the contaminated sample was an authentic or counterfeit product, whether the seal on the bottle was broken or intact, whether the product was produced in a controlled environment and whether some kind of cross-contamination had occurred.
According to Johnson & Johnson, the FDA’s most recent test of the company’s baby powder in September along with thousands of tests conducted for more than 40 years have all found no asbestos.
In contrast, Reuters and New York Times investigations both published on December 14, 2018, found that Johnson & Johnson had known about potential asbestos contamination in its baby powder since the early 1970s but failed to warn consumers.
As of mid-afternoon following the morning announcement, Johnson & Johnson shares had fallen $8.05, about 6 percent, to $128, the biggest one-day drop in 10 months.
In recent months, we've worked to collaborate on behalf of consumer product safety. Help us put the #TruthOnTalc by signing our petition demanding Johnson & Johnson add an adequate warning label.
Sign HereFootnotes